|
11/14/2023 0 Comments Infrared spectrum chart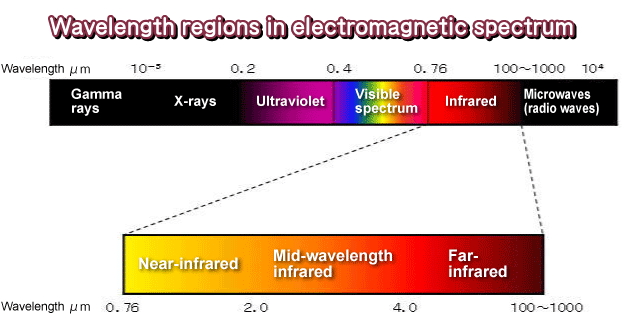
Consequently, the vibrational energy levels of sample molecules are transferred from the ground state to the excited state. The infrared spectrum is nothing but a molecular vibrational spectrum that when exposed to infrared radiation sample molecules selectively absorb radiation of specific wavelengths causing a change in the dipole moment of sample molecules. Infrared absorption spectroscopy is the method to determine the structures of molecules with the molecule’s characteristic absorption of infrared radiation. IR Spectroscopy detects frequencies of infrared light that are absorbed by a molecule and this absorption takes place due to the ability of molecular bonds that are corresponding to the specific frequency. An illustration highlighting the different regions that light can be classified into is shown in Fig-1. The major use of infrared spectroscopy is to determine the functional groups of molecules, relevant to both organic and inorganic chemistry An IR spectrum is essentially a graph plotted with the frequency or wavelength on the X-axis and infrared light absorbed on the Y-axis. The concept of IR spectroscopy can be generally analyzed in three different ways first one by measuring reflection, second by emission, and third by absorption. The range of Infrared region is 12800 ~ 10 cm-1 and can be divided into near-infrared region (12800 ~ 4000 cm-1), mid-infrared region (4000 ~ 200 cm-1) and far-infrared region (50 ~ 1000 cm-1). Infrared Spectroscopy is a technique for analyzing the interaction of molecules with infrared light. Infrared light was discovered in the 19 th century which led to the use of infrared lights in various applications. 
Infrared (IR) spectroscopy deals with the infrared region of the electromagnetic spectrum, i.e., light having a longer wavelength and a lower frequency than visible light.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
